The Syn-Fill™ Line of Filling Solutions
Phaser Solutions revolutionizes the pharmaceutical and biopharmaceutical industry with its innovative filling equipment and consumables. Our cutting-edge line of single-use manifold technologies for use with Prefilled Syringes, Vials, and Bags empowers companies to streamline their filling operations, ensuring compliance and robustness while significantly reducing capital expenditure. By choosing Phaser, you’ll benefit from:
- Simplified processes: Our advanced solutions make complex filling operations more straightforward and efficient.
- Faster setup: Implement our systems quickly, allowing you to accelerate your production timelines.
- Cost-effectiveness: Enjoy lower initial investments and ongoing operational costs.
- Reduced expertise requirements: Our user-friendly technologies minimize the need for specialized knowledge.
- Better technical support: Get the support you need quickly and efficiently to get up and running
- Customized Solutions: Work with our design team on a bespoke system to maximize the benefits for your application
Experience the next generation of small batch filling with Phaser Solutions – where innovation meets efficiency in pharmaceutical manufacturing.


Product Benefits
Syn-Fill™ reduces implementation time through eliminating many of the typical planning, permitting, and construction steps associated with Isolator Filler systems. Because of this reduced timeline, having internal production capacity for robust filling operations is now a viable option in the timeline you need for pre-clinical, clinical, and commercial production facilities.

Implementation of an Isolator or RABS (Restricted Access Barrier) based filling line is typically a construction project that requires careful planning and execution. This is because most filling systems:
- Are too large to move through existing corridors and doorways in the facility, and must have a hole cut in the wall or roof of the facility to get the equipment inside or they require substantial re-assembly upon delivery
- Require construction of a dedicated suite and support areas
- Require additional utilities that directly (e.g. HVAC, VHP safety) or indirectly (e.g. WFI/PW for parts washing, Clean Steam for Autoclave) support the isolator
By eliminating these needs, the Syn-Fill™ system eliminates these added costs, leaving only the running of new electrical circuits, equipment costs, and CQV. Because of this, typical end users will be able to save more than $1M in capital costs.
3 design elements put phaser in the lead for throughput:
- Pre-cleaned and sterilized closed process manifold
- Using pre-cleaned and sterilized process contact flow paths reduces setup and post run cleanup timelines, which can save hours to days of time in getting ready for a fill.
- Fully assembled containers
- Your final containers come fully assembled with a plunger/stopper, crimp cap/plunger rod & bellows, and need only be filled using the manifold tubing and an automated process. This eliminates process steps and potential bottle necks that can be caused by the washer, depyrogenation tunnel, filler, stopper, capper and decontamination vestibules.
- Synchronous filling
- Filler system speed is largely dictated by the serial (one after another) processing design of the systems. To reach the high throughput rates needed for filling of time/environment sensitive materials additional needles are employed. With a Syn-Fill™ system all filling is synchronous whether it is 20 units or 2,000 units. Because of this approach the syn-fill is unmatched in processing time even when compared with multi needle high speed filling systems.
Open filling of syringes or vials has the inherent risk that the environment will contaminate the interior of the container and adulterate the product, making it unsafe for use. Once the units are filled and closed, the environment they are in is no longer a risk and they can be removed from the classified cleanrooms and shipped through all sorts of environments without risk to the product. The Syn-Fill™ system starts with a closed system and uses a sterile connection to enable filling through the manifold. By doing this, we enable safe, sterile, robust filling without the need for classified cleanrooms.
By creating a closed process flowpath that is integrity tested before and after filling, the Syn-Fill™ system may eliminate the need for:
- Isolator
- Dedicated suite
- Material air-locks
- Personnel air-locks
- Classified corridors
- Locker rooms
- Parts wash suites
- Parts washers
- Autoclaves
- Support utilities
Utilizing a compact design, the base model takes only 3’ x 8’ (1m x 2.4m) of floor space and can fit through a standard door frame. Combine multiple units to scale up operations or swap them out to meet the varied needs of a flexible manufacturing space.
An isolator filler and associated equipment are a complicated set of systems often requiring disparate expertise / knowledge from specialized process engineers, facilities engineers, automation engineers, quality assurance, regulatory, and others. The Syn-Fill™ system doesn’t require the same level of expertise or time commitment from other departments. This is primarily due to the premade assemblies, simple operations, reduced need for cleanroom and utility systems, and small footprint. In addition, Phaser has internal experts and has partnered with reputable engineering service providers to ensure you receive the support needed for such a critical and often complicated process.
With a pre-sterilized modular filling manifold and fully automated filling, you may eliminate or reduce:
- Containment system cleaning
- Containment system sterilization
- Environmental monitoring during fill
- HVAC maintenance
- EM trending
- Container washing
- Container depyrogenation
- Periodic requalification of cleaning, sterilization, and depyrogenation
- Complex automated assembly of fill containers
- Operator aseptic technique qualification
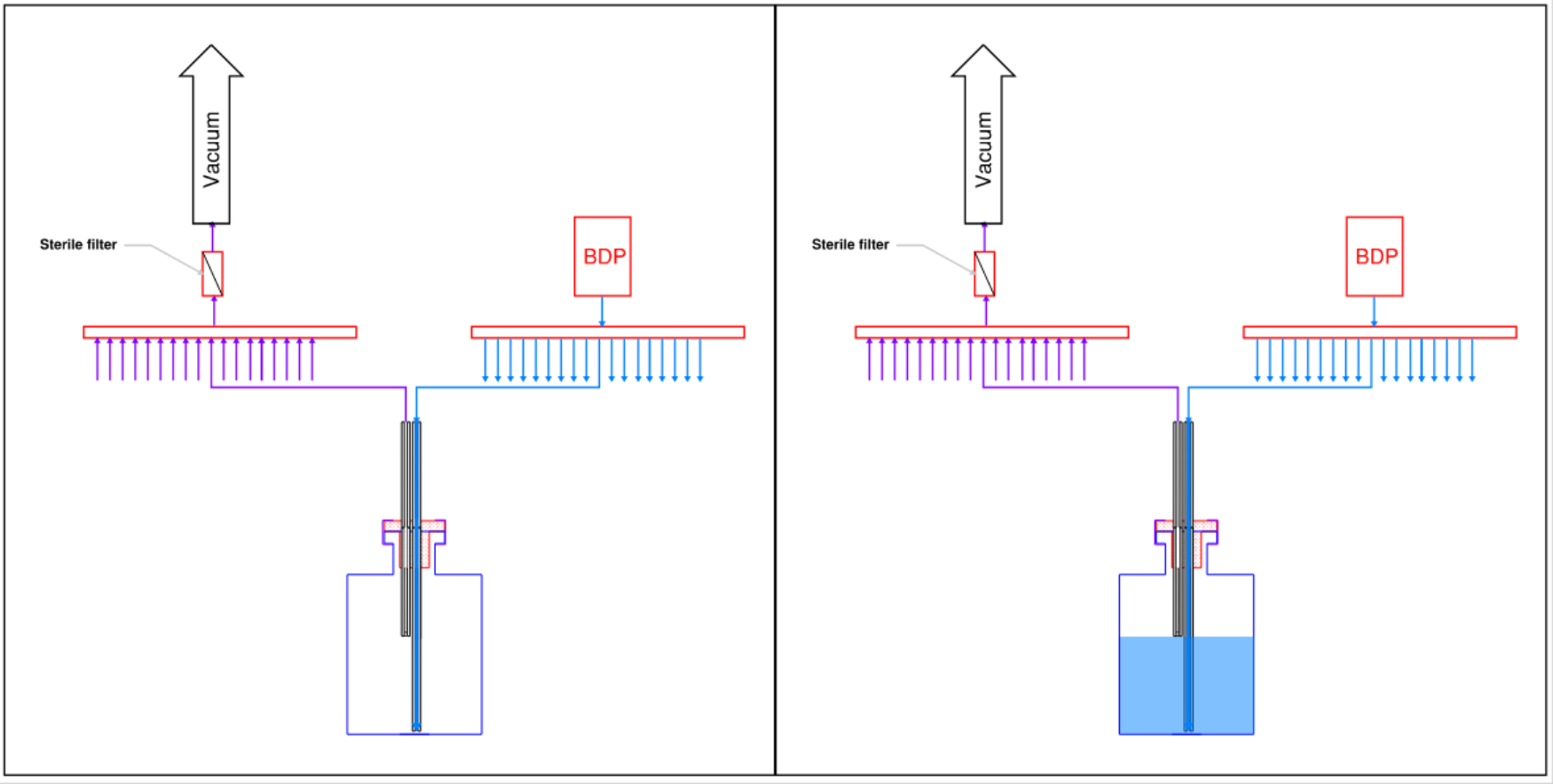
Many of Today’s filler systems require priming of the filling line by flushing product from the bulk through the filling needle(s) and into a waste container. This is necessary to remove air bubbles from the line and ensure filled vials have the correct fill volume. Syn-Fill™ systems have been re-envisioned to maximize your filling process yield by eliminating this product loss. For syringes and bags, we perform an initial filling of the containers in their upright orientation, which allows any air in the containers to rise to the top due to buoyancy. The containers are then evacuated back to the bulk drug product (BDP) bag, where the bubbles again rise to the top of the container due to buoyancy. This back-and-forth step primes the line to the final containers without any product losses. After priming, the final containers are filled to their target volume and are sealed off for inspection, labeling, and packaging.
For vials, the filling approach is a bit different but achieves the same result. Each vial is provided with a special stopper that contains in inlet diptube and a vent tube that protrudes into the vial. The dip tube extends to the bottom of the vial to minimize splashing. The vent tube contains a hydrophobic membrane at the tip that prevents liquid from entering, and the length of the protrusion into the vial matches the target liquid level. A gentle and adjustable vacuum is pulled on the vent tube, which draws in bulk drug product from the header. Liquid will continue to flow into the vial until it reaches the hydrophobic filter on the vent tube, at which point the liquid cannot continue to flow.
Vials are a tried-and-true technology for containment that work under a multitude of storage conditions. Because of their robustness they have become the go-to solution for primary containment. However, they have the drawback of needing to have a 10-30% overfill to ensure the clinician attempting to extract the filled product into a syringe can easily get the required dosage. The alternative is to fill into a syringe where overfilling can be significantly reduced because the therapeutic is already in the syringe and ready for administration. However, syringes are not without their drawbacks. The primary drawback is that the container closure is less robust because of the moveable plunger in the barrel, which can potentially allow for microbial ingress or product leakage.
The Syn-Fill™ line of filling options offers syringe filling that combines the best aspects of both a syringe and a vial. To enhance the container closure integrity, Syn-Fill™ uses a bellows on the plunger rod, providing additional protection for the non-product side of the plunger, and offers plungers from a variety of vendors which can meet the specific storage and transportation requirements of most applications. As such, a drug which otherwise would have required a vial for storage and transportation can be filled into syringes.
In addition to yield improvements from filling into syringes, the syn fill can enable improvements in yield through Zero Loss Priming and Reduced QC Sampling losses
When using the Syn-Fill™ system to fill into syringes or bags, the manifold degassing is performed by filling and emptying the containers from/to the bulk drug product bag. When this happens, it degasses the manifold and performs mixing. If desired, a similar operation can be implemented for vial filling by reversing the vacuum to a low-pressure overlay until all product is returned to the drug product bag, and then by pulling vacuum again to perform the final filling of the product. This in-process mixing combined with the synchronous short duration filling ensures the product is mixed and doesn’t have time for developing concentration gradients during the filling. This post setup mixing and fast filling reduces the risk inherent in legacy filling systems which can have product settle out or degrade throughout the fill due to longer setup times, no in-process mixing, and filling in series .
The Syn-Fill™ system requires very few utilities:
Requires:
- 208/240V power
- Compressed Gas (e.g. CDA, Helium, Argon, Nitrogen)
Not Required
- Purified water
- Clean steam
- HVAC
- CIP
- Chilled water/glycol
The 208/240V power source is required for running the automation and performing heat sealing/cutting
The compressed gas is required for performing integrity testing and actuation of the sealing/cutting heads. Compressed gas consumption rates are relatively low and can typically be achieved through use of a bottled gas.
The Syn-Fill™ system can operate inside of a cold room or other controlled temperature environment to ensure temperature sensitive therapeutics have the protection they need. Additionally, the incredibly fast cycle times (<2hr/batch) reduce the time the product needs to be outside its storage conditions.
For parenteral preparations, USP <71> requires the sampling to be representative of the entire batch to ensure uniformity. To meet this requirement, samples are typically collected from the Beginning, Middle, and End of each fill, and after significant process interventions. With the Syn-Fill, all filled units are filled synchronously over a short period of time so there is less chance of something happening in the course of the filling operations that could alter the uniformity (e.g. change in environmental contaminants, settling of product in the bulk container, changes in pump dispensing mass/volume). As a result of the novel filling mechanism, the Syn-Fill™ system reduces the chances of variance in uniformity and thus the need for redundant sampling and IPC controls to verify the uniformity.
Syn-Fill™ systems can reduce your Commissioning, Qualification, and Validation (CQV) requirements.
Equipment Qualification
The Syn-Fill™ system’s approach to filling doesn’t rely on external utilities systems and movement of the final containers through a series of stations requiring multi-axis movement arms, conveyors, weigh stations, pumps, vision systems, and an automation system conducting a symphony of actions. As a result of the simplicity of the system, the automation and qualification are also simplified. This enables faster and less expensive qualification for implementation.
Environmental Qualification
Because the Syn-Fill™ utilizes closed system processing, it can be operated in reduced cleanroom classifications (Refer to ICH Q7, PIC/s “Guide to Good Manufacturing Practice for Medical Products (Part II), BioPhorum, Process Closure Playbook). This reduces the qualification needs for the environment in which the filling equipment will be operated.
Cleaning and Sterilization
The Syn-Fill™ system uses a completely disposable process contact flowpath. No cleaning and no sterilization are required. We take care of all of that, sending you the final containers as part of a modular manifold system that is ready to process as soon as it is installed, connected, and passes integrity testing. There is no need for assessing manual or automated cleaning, no need for:
- Isolator / RABS sterilization/sanitization validation
- Container washing & drying
- Depyrogenation
and no need to perform periodic re-qualification or validation of these critical aspects.
With the simplicity of system implementation, functionality, and operator use; fewer personnel with less overall isolator and cleanroom specific knowledge are needed due to the simplicity of Syn-Fill™ system implementation, functionality, and operator use. Implementation and operation of a Syn-Fill™ system is comparable in complexity to a single use chromatography system or bioreactor; requiring input from multiple departments but predominantly completed by a single lead engineer. Comparatively, the implementation of an isolator filler system may require a wider breadth of knowledge and is typically supported using external consultants for the project. This may include:
- Facilities Design/Engineering
- Process Design/Engineering
- Sterile product manufacturing regulatory expertise
- Sterile product manufacturing QC expertise (Samples & Environmental Monitoring)
- Isolator/Filler CQV expertise
- Architectural design of sterile product manufacturing areas
- Project management
Operation of the Syn-Fill™ system requires less personnel as well as a result of: - No personnel needed for cleaning and sterilization of the system (only for operations)
- No isolator/RABS filler engineering staff needed for setup and maintenance of complex robotics, conveyor, HVAC, and sterilization systems
- Reduced environmental classification and closed processing, which reduces the personnel needed for environmental monitoring, sample processing, deviation investigations, and trend report generation.
See how Syn-Fill™ impacts stakeholders
